[category science-report]
END-MISSION SCIENCE REPORT – G. GÉGO – MDRS 286
Introduction
The CO2PROT project aims to develop an efficient, sustainable and reliable Bacteriological Life Support System for manned space exploration using purple bacteria.
Purple bacteria are known for their metabolic heterogeneity, which allows for different compounds, like wastes or in situ resources, to be envisaged as substrates.
Among these, carbon dioxide remediation is by far the most attractive option, as it traps waste into potentially edible biomass. With the carbon source defined, multiple electron sources are available, but no comparative data has ever been accumulated to rule out the better option, would it be for space exploration or terrestrial applications.
In this study, three main metabolisms leading to CO2 fixation will be compared by studying the growth of purple bacteria model Rhodospirillum rubrum in:
Photoheterotrophy: High-electron-content volatile fatty acids (Butyrate/Valerate).
Photoautohydrogenotrophy: Hydrogen.
Photoautoelectrotrophy: Electron flux (current).
The bacteria will be grown inside low-cost bag photobioreactors to assess the possibility of mass-production in altered gravity, while reducing costs of terrestrial downfalls of the study. Analog missions are therefore ideal platforms to test if such installations are feasible on other planets. Since photoheterotrophy was already studied in another analog (AATC, Poland), photoautohydrogenotrophy will be tested at MDRS as a follow-up.
Figure 1: Cultivation chamber & electrolyser. Five bag bioreactors were inoculated with purple bacterium Rhodopseudomonas palustris TIE-1. The carbon source used is baking soda, and the electron source is hydrogen produced via electrolysis. The bags are constantly agitated by a rocking platform, which helps solubilize the gas phase within the freshwater media. Green (525nm), Orange (592nm) and infrared (850nm) LED strips are used to supply photons to the anoxygenic photosynthesis pigments.
Methods
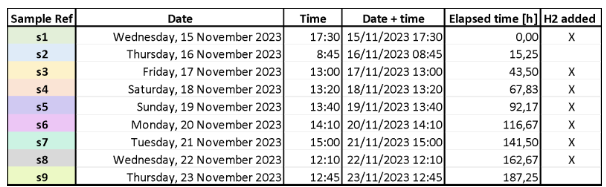
In the science dome, the 5 bags inoculated with purple bacteria Rhodopseudomonas palustris TIE-1 are continuing their growth steadily. Turbidity (optical density), measured by spectrophotometry, increased in all photobioreactors, indicating nominal conditions. One sample is taken each 24h for each bag, centrifugated, and the supernatant is separated from the pelleted bacteria, then stored at -20°C. H2 is produced using water electrolysis (300 g of NaOH in 3 L of H2O were used). H2 is supplied ad libitum daily, since the expected consumption of H2 cannot be estimated easily.
Results
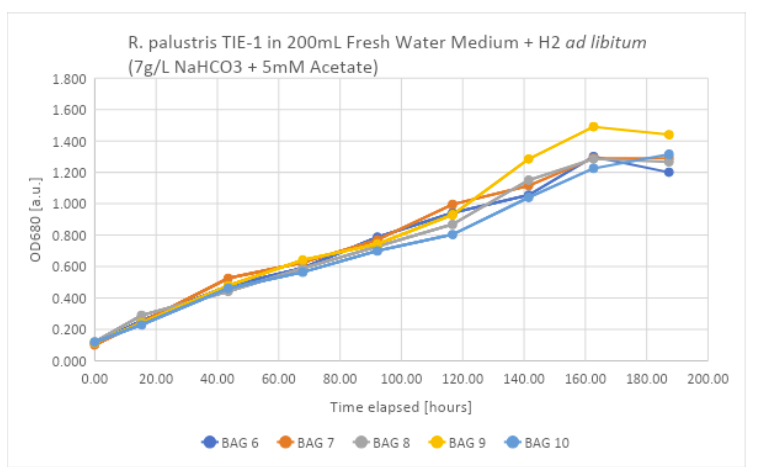
Here is a graph showing the evolution of OD (measured at 680nm) for all 5 bioreactors.
Figure 2: OD measurements between SOL 3 and 5. Growth is visible and follows known trends. Similar experiments will then be performed at the University of Mons to check the results that were obtained.
On day 9 of the experiment, the bacteria finally showed a stationary phase, indicating that the growth of the bacteria reached its peak. Final ODs oscillate around 1.2 and 1.4, with bag n°9 showing higher growth than the others. This could be due to better lighting, better agitation, or errors in measuring the OD and contamination.
Table 1: Low-cost bag photobioreactors sampling schedule.



You must be logged in to post a comment.